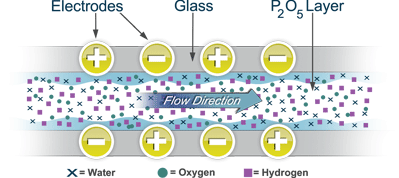
Based on Faraday’s Law of Electrolysis, the MEECO cell absorbs and electrolyzes moisture at fractional parts-per-million (ppm) or other units of measure. How: One hundred percent of the sample moisture is absorbed by a phosphorus pentoxide (P2O5) film that covers two spirally-wound electrodes embedded in a hollow glass tube. When the sample gas enters the cell at a known flow rate, the film absorbs all the moisture molecules present. By applying an electrical potential (voltage) to the electrodes, each absorbed water molecule is electrolyzed, generating a finite current. This current is precise and proportional to the amount of absorbed water. It is, therefore, an exact, direct measurement of the water vapor present in the sample gas.
Do you really need all that complicated calibration equipment the competition wants to sell you? Wouldn’t you rather keep it simple? With MEECO instrumentation, it’s as easy as Faraday’s Law of Electrolysis: Moisture in = Current Out.
MEECO’s efficient sensor design uses phosphorus pentoxide (P205), a chemical so hygroscopic — that is, spongy – it’s used to make gas dryers. Employed as a coating in MEECO sensors, P205 absorbs each and every water molecule present in the gas flow.
When you reach equilibrium – a steady state – water molecules electrolyze at a rate equivalent to the rate they enter the sensor. With each electrolyzed water molecule, two electrons jump from the anode to the cathode. In other words, it’s a linear process.
By measuring the electrolysis current, you know exactly how many water molecules are present in your gas stream. So, water in equals current out. When you set the gas flow through the cell at a known rate, the current tells the concentration of water.
There’s no need for calibration. Once you measure the current, and set it and the gas flow properly, you know the moisture concentration. There’s no need to take the unit off line or even to turn it off.
The MEECO moisture analyzer – complete and uncomplicated. It’s simply all you need.